Severe bleeding is one of the leading preventable causes of death in trauma care, and the body’s natural response — blood clotting — is remarkable but slow. Clots take minutes to form and minutes to mature, and even when they do, they tend to be mechanically fragile, prone to fracturing under pressure or shifting tissue. For years, researchers have tried to design biomaterials that can substitute or reinforce this process: hemostatic powders, hydrogels, surgical sealants. Most rely on reactions that ignore the blood itself. A study published this week in Nature from a team at McGill University takes a different route. It uses the blood.
The technique is called click clotting. Led by Jianyu Li at McGill’s Department of Mechanical Engineering, with first author Shuaibing Jiang (now at Harvard Medical School and Mass General Brigham), the team showed that red blood cells — which most people think of only as oxygen carriers, not structural material — can be turned into the load-bearing component of a clot if you connect them to each other the right way.
Linking cells with chemistry
The connection method is borrowed from synthetic chemistry. “Click reactions” are a class of fast, selective chemical reactions that snap two specific molecular handles together with high efficiency and minimal side products. They’ve been used in drug development and protein labeling for years. The McGill team modified red blood cells so that surface proteins on neighboring cells could click together — forming a covalent bond that locks the cells into a connected gel structure.
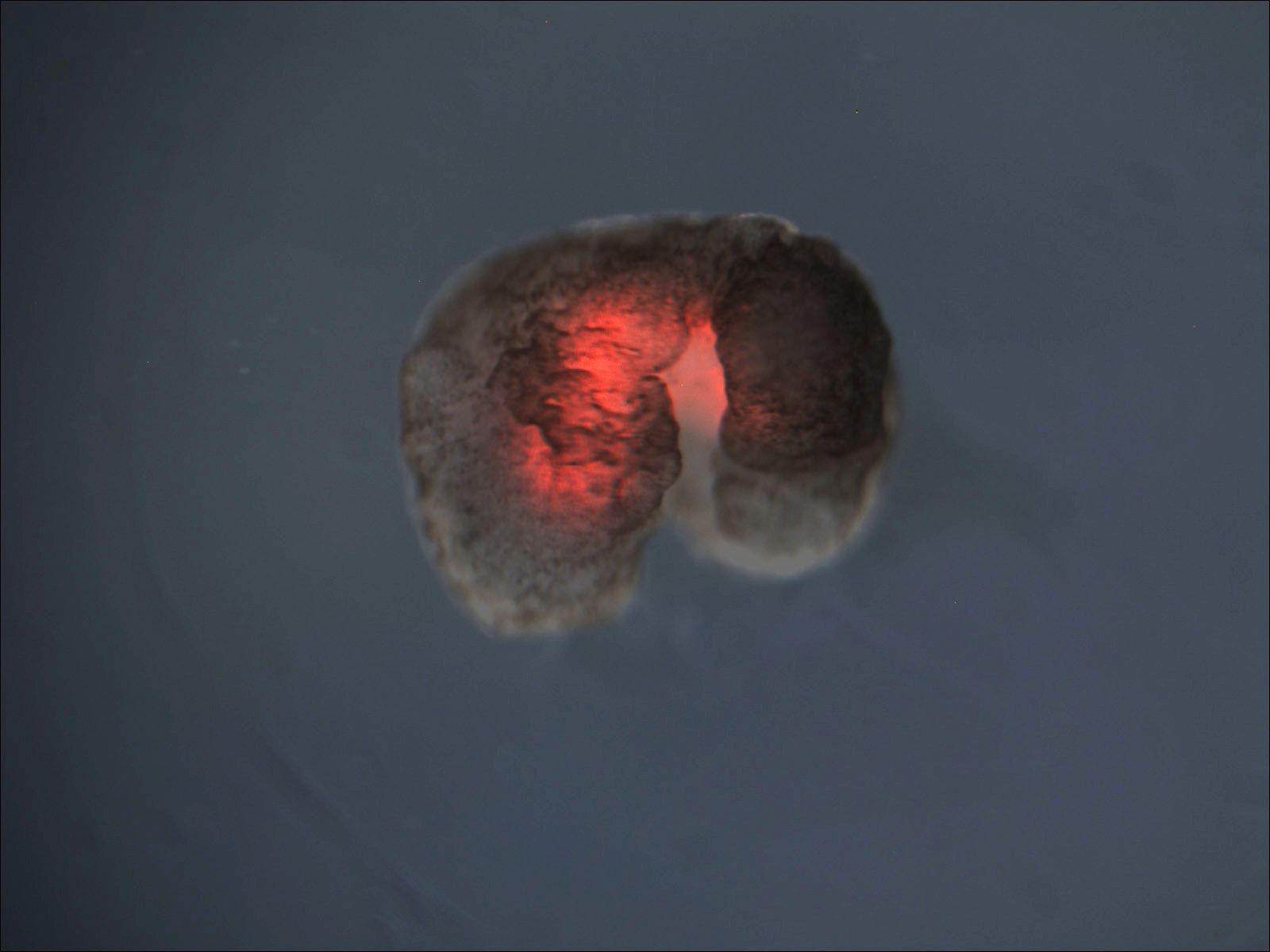
The result is what they call a cytogel. Once added to whole blood, the cytogel gels in roughly five seconds and becomes embedded in the body’s natural fibrin network. Mechanical testing showed the engineered clots were thirteen times more resistant to fracturing and four times more adhesive than natural ones. Earlier attempts to crosslink red blood cells using chitosan, a polymer derived from crustacean shells, had run into the same wall: brittle clots, ruptured cells, and inconsistent performance. The click reaction sidesteps that, partly because it doesn’t disrupt the cell membrane and partly because it doesn’t interfere with normal blood chemistry.
Tested in injured rats
The researchers tested the cytogel in vitro and in rat models, including a severe liver laceration. The engineered clots stopped bleeding faster and supported tissue regeneration better than a clinically used hemostatic product included as a comparison. Histology after 28 days showed minimal inflammation, normal tissue architecture, and no signs of toxicity in major organs. The team also showed the system works for both autologous applications — using a patient’s own blood, with about a 20-minute prep time — and allogeneic ones, using type-matched donor blood, prepared in around 10 minutes.
The pace matters because trauma medicine is constrained by minutes. Most existing hemostatic products either need to be applied directly to a wound surface and held there, or rely on coagulation factors that don’t function well in patients with clotting disorders. A pre-formed, ready-to-deploy cytogel that integrates with the body’s natural fibrin clot offers a different kind of flexibility.
What the work doesn’t yet show
The study is in rodents and in vitro, not in people. Translating biomaterials from rat liver lacerations to human emergency care is a long road that involves scale, manufacturing, regulatory review, and testing across a much wider range of injury types. There are also open questions about how long the cytogel persists in tissue, how the body breaks it down over time, and whether the chemistry stays stable across different patient populations.
For now, the contribution is both conceptual and concrete. Conceptually, it reframes red blood cells as more than oxygen carriers — they can serve as the structural unit of a designed material. Concretely, it shows that a clot built this way is faster, tougher, and more adhesive than what the body produces on its own, while staying biocompatible enough to support healing rather than just seal the wound.